 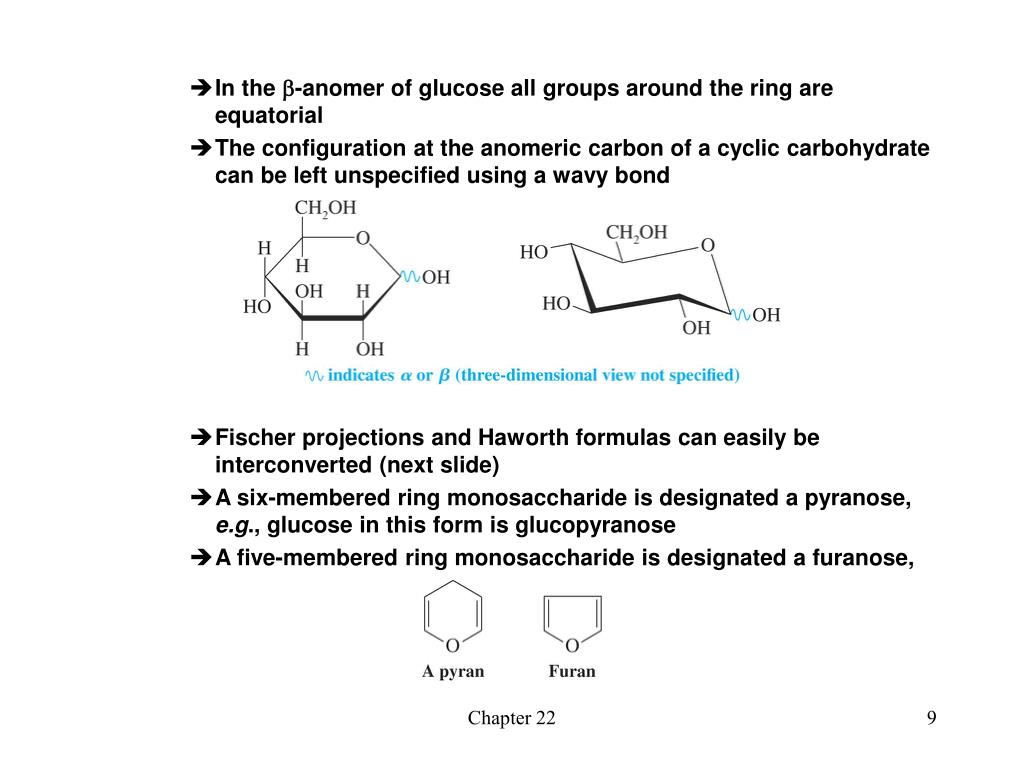
We refer to such stereoisomers as diastereomers. Some of these stereoisomers will have enantiomeric relationships, but enantiomers come in pairs, and non-enantiomeric stereoisomers will therefore be common. 
Depending on the overall symmetry of the molecular structure, some of these combinations may be identical, but in the absence of such identity, we would expect to find 2 n stereoisomers. These are shown in the following illustration, together with the assignments that have been made on the basis of chemical interconversions.Īs a general rule, a structure having n chiral centers will have 2 n possible combinations of these centers. Each may assume an R or S configuration, so there are four stereoisomeric combinations possible. The structural formula of 2-methylamino-1-phenylpropanol has two stereogenic carbons (#1 & #2). Although these missing stereoisomers were not present in the natural source, they have been prepared synthetically and have the expected identical physical properties and opposite-sign specific rotations with those listed above. Since these two compounds are optically active, each must have an enantiomer. 119 º C, D = +52º, relatively insoluble in water 35 - 40 º C, D = –41º, moderate water solubility How, then, are we to classify these isomers and others like them? Since the properties of these compounds (see below) are significantly different, they cannot be enantiomers. Both compounds are stereoisomers of 2-methylamino-1-phenyl-1-propanol, and both are optically active, one being levorotatory and the other dextrorotatory. The Chinese shrub Ma Huang ( Ephedra vulgaris) contains two physiologically active compounds ephedrine and pseudoephedrine. Note that the R/S assignment is unchanged.Ĭompounds Having Two or More Chiral Centers For an example, click on the diagram to see the 1:3:4-face, shaded light gray. Once one recognizes this relationship, the viewing options are increased and a configurational assignment is more easily achieved. Now a tetrahedral structure may be viewed from any of the four triangular faces, and the symmetry of the system is such that a correct R/S assignment is made if the remote out-of plane group has an even number sequence priority (2 or 4), whereas the wrong assignment results when the out-of plane group has an odd priority (1 or 3). The viewing rule states that when the lowest priority substituent (4) is oriented behind the triangular face defined by the three higher priority substituents (shaded light gray here), a clockwise sequential arrangement of these substituents (1, 2 & 3) is defined as R, and a counter-clockwise sequence as S. The higher the atomic number of the immediate substituent atom, the higher the priority.įor example, H– 2 > 3 > 4), then the R and S configurations are defined. The Sequence Rule for Assignment of Configurations to Chiral CentersĪssign sequence priorities to the four substituents by looking at the atoms attached directly to the chiral center.ġ. Since most of the chiral stereogenic centers we shall encounter are asymmetric carbons, all four different substituents must be ordered in this fashion. 
The sequence rule is the same as that used for assigning E-Z prefixes to double bond stereoisomers. The assignment of these prefixes depends on the application of two rules: The Sequence Rule and The Viewing Rule. The symbol R comes from the Latin rectus for right, and S from the Latin sinister for left. No chemical reactions or interrelationship are required for this assignment. In the CIP system of nomenclature, each chiral center in a molecule is assigned a prefix (R or S), according to whether its configuration is right- or left-handed. The resulting nomenclature system is sometimes called the CIP system or the R-S system. A final solution to the vexing problem of configuration assignment was devised by three European chemists: R. Although this notation is still applied to carbohydrates and amino acids, it required chemical transformations to establish group relationships, and proved to be ambiguous in its general application. An early procedure assigned a D prefix to enantiomers chemically related to a right-handed reference compound and a L prefix to a similarly related left-handed group of enantiomers. We have referred to the mirror-image configurations of enantiomers as "right-handed" and "left-handed", but deciding which is which is not a trivial task. Designating the Configuration of Chiral CentersĪlthough enantiomers may be identified by their characteristic specific rotations, the assignment of a unique configuration to each has not yet been discussed. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
